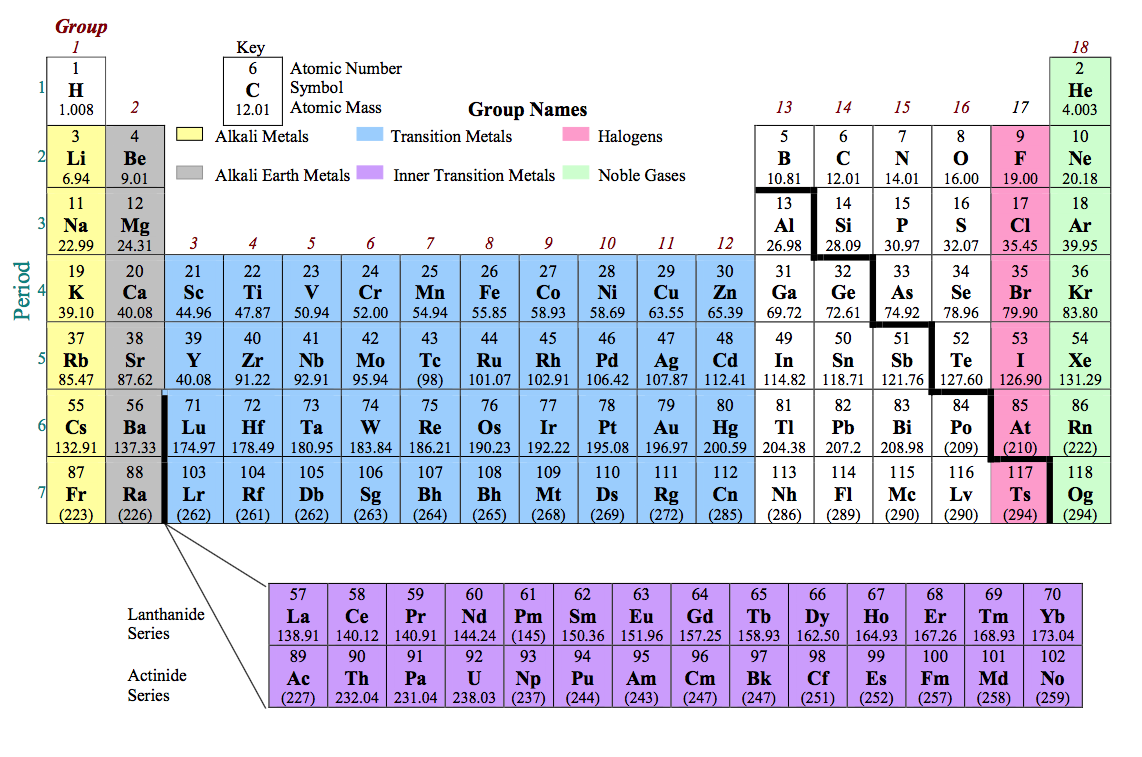
The electronic configuration of Sodium will be 1s2 2s2 2p6 3s1. How do you write the electron configuration for Sodium? What is the electronic configuration of Sodium 11? What is the boiling Point of Sodium in Kelvin?īoiling Point of Sodium in Kelvin is 1156 K. It is in the subgroup of alkaline metals NaO sodium oxide and NaOH sodium. Melting Point of Sodium in Kelvin is 370.87 K. Sodium is an element in the 1 group of the 3 period of the Periodic Table. What is the melting Point of Sodium in Kelvin? What is the boiling Point of Sodium?īoiling Point of Sodium is 1156 K. Sodium has 11 electrons out of which 1 valence electrons are present in the 3s1 outer orbitals of atom. For example, most noble gases have names ending with -on, while most. Atomic Number: 11: Element Symbol: Na: Element Name: Sodium: Atomic Weight: 22.989770: Group Number: 1: Group Name: Alkali metal: Period Number: 3: Block: s-block. The names of some elements indicate their element group. If there is a second letter, it is lowercase. Each element has a symbol, which is one or two letters. How many valence electrons does a Sodium atom have? The periodic table lists the elements in order of increasing atomic number. Sodium is an alkali metal, with atomic number 11 in Group 1 Ia of the periodic table. It is located in group 1 and period 3 in the modern periodic table. Sodium is the 11 element on the periodic table. Sodium is a chemical element with the symbol Na and atomic number 11. What is the position of Sodium in the Periodic Table? Sodium is a chemical element with symbol Na and atomic number 11. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Sodium is 3s1. 
What is the abbreviated electronic configuration of Sodium? The electronic configuration of Sodium is 1s2 2s2 2p6 3s1. What is the electronic configuration of Sodium? ANDRITZ will equip the power plant with a 170-megawatt variable-speed reversible pump turbine, the generator and related automation. 
Sodium Thermal Properties - Enthalpies and thermodynamics Li, Na, K, Rb, and Cs are all group IA elements, also known as the alkali metals. Electronelectron repulsion is important in fluorine because of its small atomic volume, making the electron affinity of fluorine less than that of chlorine. Optical Properties of Sodium Refractive IndexĪcoustic Properties of Sodium Speed of Sound Only the electron affinity and the bond dissociation energy of fluorine differ significantly from the expected periodic trends shown in Table 18.13.1 18.13. Sodium Heat and Conduction Properties Thermal Conductivity Refer to table below for the Electrical properties ofSodium Electrical Conductivity Sodium Na Sodium 11 22.990 Fact box Uses and properties History Atomic data Oxidation states and isotopes Supply risk Pressure and temperature data advanced Podcasts Video References Explore all elements Element Sodium (Na), Group 1, Atomic Number 11, s-block, Mass 22.990. Hardness of Sodium - Tests to Measure of Hardness of Element Mohs Hardness Any group whose order is a square of a prime number is also abelian.Refer to below table for Sodium Physical Properties DensityĠ.968 g/cm3(when liquid at m.p density is $0.927 g/cm3) group, and the element was named in his honor. The theory had been first developed in the 1879 paper of Georg Frobenius and Ludwig Stickelberger and later was both simplified and generalized to finitely generated modules over a principal ideal domain, forming an important chapter of linear algebra.Īny group of prime order is isomorphic to a cyclic group and therefore abelian. Sodium, like every reactive element, is never found free in nature. Sodium is an alkali metal, being in group 1 of the periodic table. It is a soft, silvery-white, highly reactive metal. The automorphism group of a finite abelian group can be described directly in terms of these invariants. Sodium is a chemical element with the symbol Na (from Latin natrium) and atomic number 11. It turns out that an arbitrary finite abelian group is isomorphic to a direct sum of finite cyclic groups of prime power order, and these orders are uniquely determined, forming a complete system of invariants. An abelian group is a set A, were among the first examples of groups.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
